Although there’s a mountain of candidates, that’s not to say there haven’t been any promising leads. Earlier this year a team led by the Liverpool School of Tropical Medicine, and including chemists from Imperial College London, announced the discovery of a fast-acting, and highly effective compound. I’m not a chemist, or even a chemical biologist, but I think the drug discovery process is fascinating. The team took the initial ‘hit’ molecule and used it as a starting point to design an effective drug candidate (a process called lead optimisation). They derived a quinazoline, with a central core of two carbon rings fused together, and from this an azaquinazoline (i.e. modified to replace a carbon atom with a nitrogen atom).
The drug was modified further, including the addition of a trifluoromethyl group (see below, the side group with three fluorines and a carbon). Quinazoline derivatives such as this are often used to fight malaria and cancer; in this case the modifications made the drug more potent, and they called it AWZ1066S.
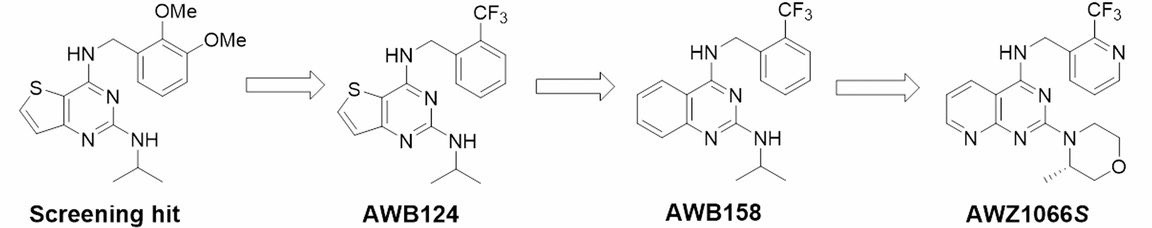
Image credit: Figure 1 from the paper by Hong et al. is licensed under CC BY-NC-ND 4.0
It meets the main criterion: it’s fast-acting, and treatment over a week leads to Wolbachia depletion of over 90%. This stops the worms from producing microfilaria, but the macrofilaricidal effect in humans is only predicted; it needs to be confirmed in clinical trials. Currently, the drug is in preclinical testing, to see if to see if it is safe for trial in humans.
Looking at the figure below, we can see that AWZ1066S is able to kill the majority of Wolbachia within just one day, an astonishing performance. The fact that it does this so much faster than other treatments suggests something profound: that it has a completely unique mode of action, a ‘first-in-class’ drug.
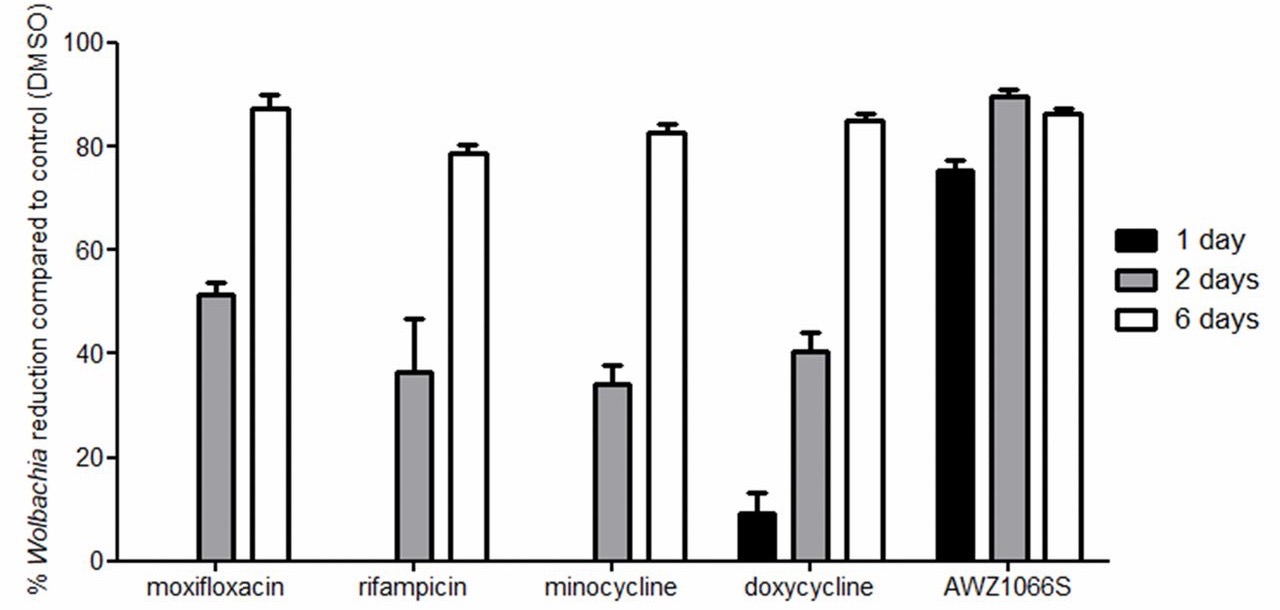
Image credit: Figure 3 from the paper by Hong et al. is licensed under CC BY-NC-ND 4.0
This means that the drug will act fast enough to thwart bacterial resistance and, perhaps more importantly, the mode of action could be adapted to fight other bacteria. The trouble is, we don’t know what AWZ1066S targets in Wolbachia.
I asked Professor Ed Tate, from the Department of Chemistry at Imperial, and who led the work there. He’s also director of the Imperial Centre for Drug Discovery Science, responsible for “nucleating critical mass within the College” in the design of novel drugs—a rather curious mission statement, but of undeniable importance. He said:
“Knowing the target might let us ‘hop’ to the same target in other bacteria. If the target is novel then this could open up new ways to treat antimicrobial resistance in general. However, target identification in Wolbachia is very challenging thanks to the limited molecular cell biology and genetic tools available to undertake.”
The team did use chemical probes to identify some potential targets of the drug, but the problem still looms large: we don’t know how it works. Prof Tate says that finding this out would help determine “the risk of off-target effects both in humans and on other bacteria, and would help us make backup candidates in case the first drug meets problems in the clinic.”
Another boon to AWZ1066S is that it’s highly selective. This is a big advantage as the drug won’t kill the friendly gut bacteria, which play important roles in humans and other mammals. For example, Clostridium difficile is a commensal found in the gut (that is, they derive a mutual benefit from their hosts). They produce a compound called butyrate, which reacts with oxygen to produce carbon dioxide, lowering the oxygen concentration in the gut and making it harder for some harmful bacteria (which rely on the oxygen) to survive.
This is illustrated below: if an antibiotic (such as doxycycline) has off-target effects, these gut commensals may well be killed off, removing this very important line of defence. In these circumstances, nasty bugs gain the upper hand leading to some unpleasant side effects.

The feature is so important, it was selected for from the start by ensuring that it killed only Wolbachia and not other common gut bacteria. However, Prof Tate said “we do not currently understand whyit is selective, because we have not yet understood the mechanism by which it works in the bacterium”. He goes on to explain that it could be due to a variety of reasons, including selective uptake and accumulation, different metabolism, or a particular version of a common protein that’s particularly susceptible to the drug in Wolbachia.

So, there you have it. A very promising look at just one of the candidates which might one day help rid the world of elephantiasis. After a shockingly long history of this disease in humans (an ancient Egyptian statue of Pharaoh Mentuhotep II, who ruled over 4000 years ago, features the characteristic swelling), we are on the brink of its elimination. On behalf of the millions of people who suffer from the disease today, I can safely say it won’t be missed.
